
The FSSAI has notified the ‘Food Safety and Standards (Approval for Non-Specified Food and Food Ingredients) Regulations, 2017 and the same have been implemented. All Food Business Operators (FBOs) who manufacture and import non-specified food and food ingredients need to submit an application in FORM – I of these regulations along with necessary documents and fee to the Food Authority as given below.
FORM – I
(Application for approval of non-specified food and food ingredient)
- Application for (Please tick √)
- Novel food or novel food Ingredients or processed with the use of novel technology
- New additives
- New processing aids including enzymes
- Articles of food and food ingredients consisting of or isolated from microorganisms, bacteria, yeast, fungi or algae
Any other non-specified food, please specify
- General information
(1) Name of the applicant
(2) Name of the authorised person
(3) Mobile No. / Phone No.
(4) E-mail – (All communications will only be made through the above email and phone number)
(5) Name of the organisation
(6) Address of the organisation/registered office
(7) Licence number, if any
(8) Nature of business
(9) Name of the food product/ ingredient/additive/enzyme/processing aid, and proprietary name, if any
(10) Justification of the name
(11) Proposed product category
(12) Source of food ingredient(s) (animal, chemical, botanical or micro-biological)
(13) In case of animal, botanical or micro-biological source, genus and species of the organism shall be mentioned
(14) Functional use
(15) Intended use
(16) Certificate of analysis from third party National Accredited Board of Laboratories (NABL) or International Laboratories Accreditation Cooperation (ILAC) recognised laboratories shall be provided. The certificate of analysis shall include physical, chemical and microbiological parameters relevant to the product along with the validated test method and other test methods, if any with references wherever applicable
(17) Manufacturing process in brief
(18) Regulatory Status
(Mention the countries where the product is permitted for direct or indirect human consumption as food. If so, provide the level and purpose of consumption by the consumers with the relevant regulations along with the documentary evidence.)
(19) Copy of agreement of relationship of applicant and manufacturer and other entities involved in the food business of the proposed product, namely, marketer, importer, re-packer
(20) Safety Information (Documents on risk assessment or toxicity studies)
- The information shall be based on safety or risk assessment review from published studies and safety studies conducted on the ingredient and food product by the applicant
- Provide evidence to demonstrate that the proposed product or the ingredient will not adversely affect any specific population groups that is pregnant women, lactating mothers, children, elderly or any other vulnerable group
- Additional specific information
(I) Novel food or novel food Ingredients or food processed with the use of novel technology
(1) The target group for the said proposed food, if any
(2) Detailed composition of the product (with quantity of the ingredients and additives added in the product)
Any new ingredient(s) (Please specify if the product has one or more new ingredients which as on date is not listed in Food Safety and Standards Regulation, or an ingredient which has been introduced for the first time in India)
(3) Details of new technology
(4) Safety Information (Documents on risk assessment or toxicity studies to be attached)
(a) Information on human studies including dietary exposure, nutritional impact and potential impact on the consumer if any
(b) Toxicological studies including results of Ame’s tests to test mutagenicity, chromosomal aberration tests, studies for reproductive toxicity, prenatal developmental toxicity studies
(c) Allergenicity (published or unpublished reports of allergenicity or other adverse effects in humans associated with the food consumption; may include reports prepared by World Health Organisation or by other national or international agencies responsible for food safety or public health)
(5) History of consumption of food product/food ingredient, (attach supporting documents)
(a) Geographical area of use (with established history of safe use in at least two countries, with well-established regulatory status)
(b) Quantity of consumption
(c) Duration of consumption (in years)
(II) New additives
(1) Chemical name and INS No.
(2) Purity (food grade or equivalent)
(3) Acceptable Daily Intake (ADI) specified by JECFA or any other risk assessment body
(4) Proposed level of use in food category
(5) In case of colouring agent provide (Colour Index) colour number, where applicable
(III) New processing aids including enzymes
(1) Specification
(2) Enzyme activity
(3) Purity
(4) Residual limit in the final product (in case of processing aid)
(IV) Articles of food and food ingredients consisting of or isolated from microorganisms, bacteria, yeast, fungi or algae
Note: (1) Mention “not applicable” (NA) wherever necessary.
(2) All data documentary evidence provided by the applicant shall be from international peer reviewed journals, international bodies including World Health Organization (WHO) and Food and Agricultural Organization (FAO). Only complete records or studies shall be provided.
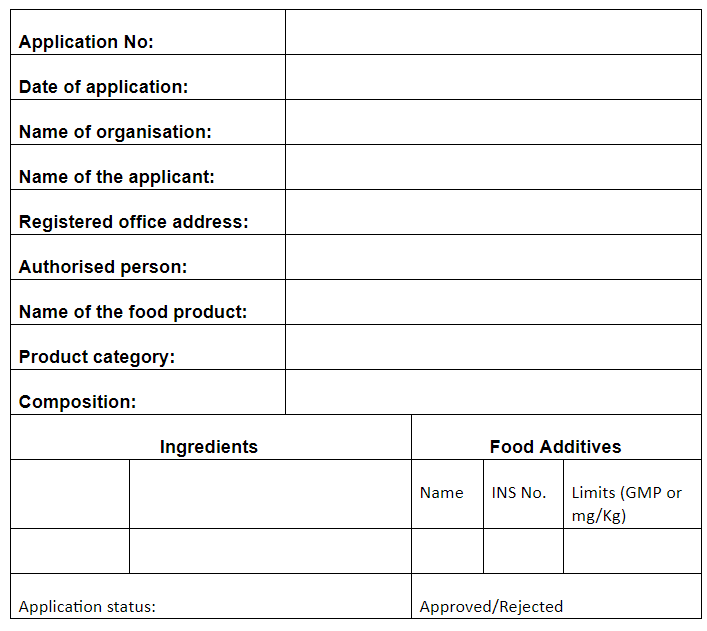
The Food Authority may either grant approval or reject the application, as per FORM-II, on the basis of the safety assessment of the article of food which is as follows
- Conditions for approval:
- Reasons for rejection, if any:
(Authorised Signatory)
Source: FSSAI

Leave a Reply