
The FSSAI has issued a Gazette Notification dated 10 September, 2021 on Food Safety and Standards (Labelling and Display) First Amendment Regulations, 2021 with regard to labelling of Multi-source Edible Oil and revision of labelling requirements for sweeteners.
The copies of the said two Gazette Notifications were made available to the public on the
18th September, 2020 and 18th November, 2020, respectively and after taking into consideration the objections and suggestions received from the public in respect of the said two draft regulations, the FSSAI has now made these regulations to amend the Food Safety and Standards (Labelling and Display) Regulations, 2020.
Regulations
These regulations may be called the Food Safety and Standards (Labelling and Display) First Amendment Regulations, 2021. They shall come into force on the date of their publication in the Official Gazette and Food Business Operator shall comply with all the provisions of these regulations by 17th November, 2021.
The following amendments have been made to the Food Safety and Standards (Labelling and Display) Regulations, 2020.
Amendments in regulation 7, Mandatory Declarations
to the sub-regulation (1), under the above regulation a proviso has been added to the following
(1) Wherever, packaged food contains ingredients and /or additives as stated in schedule -II of these regulations, the same shall be prominently displayed on the label. The size of numerals and letters for the declarations/specific requirements specified in schedule -II shall not be less than 3mm based on the letter l.
“Provided that in case of food package having surface area up to 30cm2 containing caloric or non-caloric sweetener or mixture thereof, the size of numerals and letters for the declarations or specific requirements specified in Schedule -II shall not be less than 1mm based on the letter l.”
Amendments n Schedule-II, Mandatory Declarations
(1) in clause 1 to the Table the following has been included
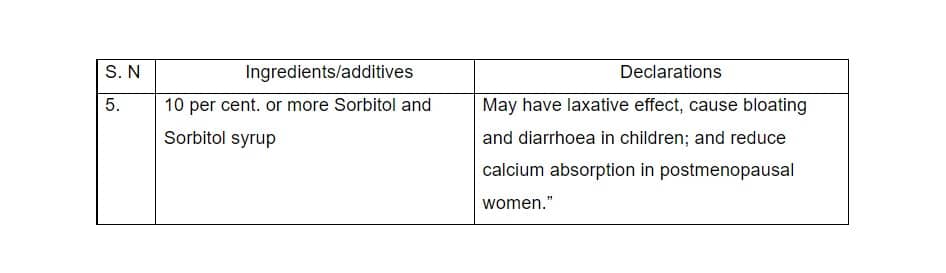
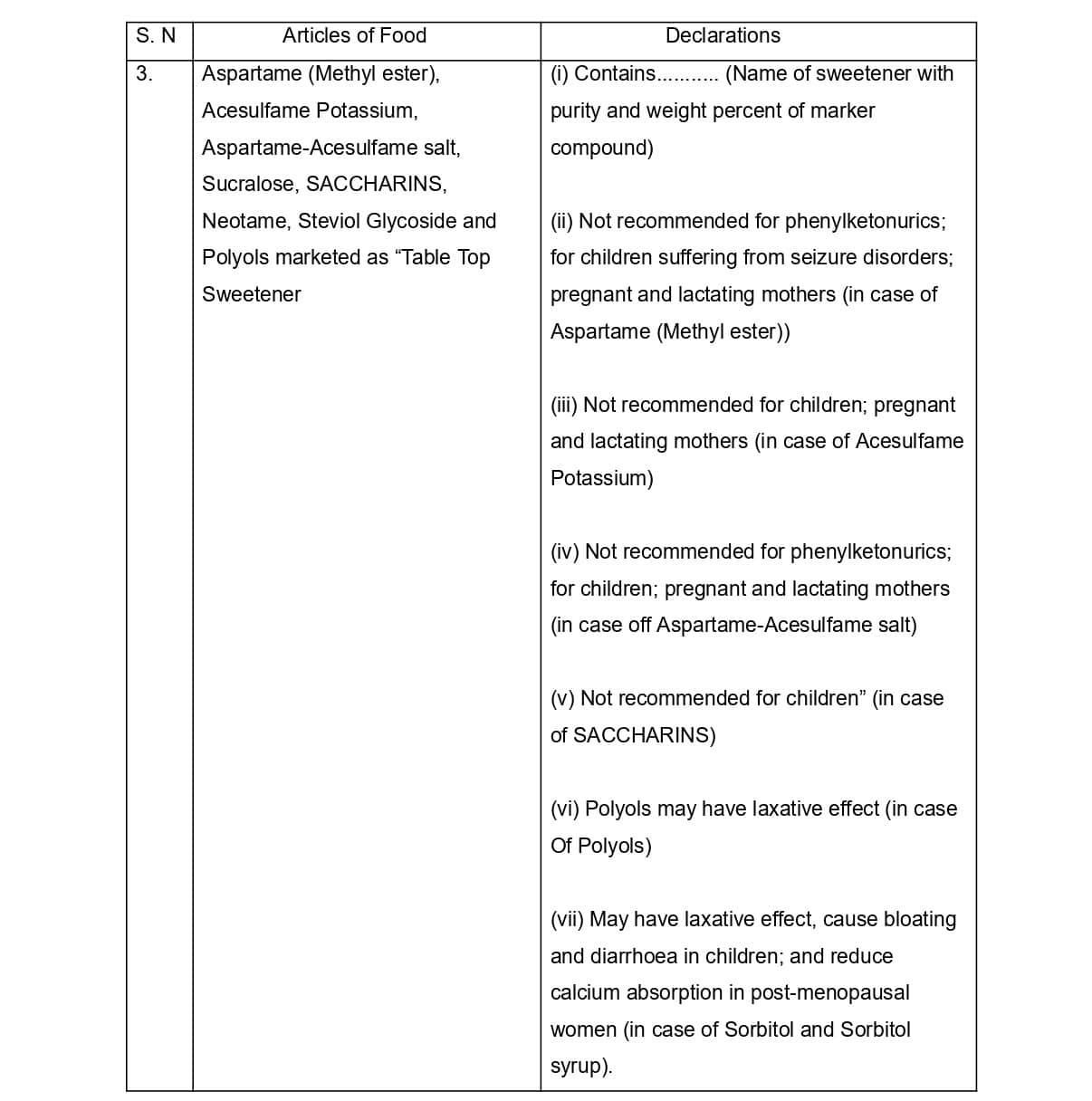
In the same Schedule in sub-clause (3) amendments have been made in serial number 3. in the table which are as follows
In the same sub- clause 3) the article of food at serial number 4. Aspartame (Methyl ester), marketed as “Table Top Sweetener” has been omitted
Amendments in Schedule II, in n sub-clause (4), the entries in the table have been changed to the following
Amendments in clause 2, relating to “Specific requirements/ restrictions on manner of labelling”,
In sub-clause 2.1 – Labelling of edible oils and fats the following is to be added
“2A. In case rice bran oil, which is physically refined is used as one of the ingredients in Vanaspati, it shall be declared in the ingredient list on the label as “Physically Refined Rice Bran Oil.”
In the same clause 2, the sub-clause (4) has been amended
“(4) Every package containing an admixture of edible oils shall carry the following label declaration in bold capital letter immediately below its brand name or trade name on the front of pack, namely: –
For pack size less than one litre, the font size of the label declaration “MULTI-SOURCE EDIBLE OIL”, shall not be less than 3 mm with the length of declaration statement as 35 mm minimum and for label declaration “Name and Nature of edible oil……per cent. by weight”, font size shall not be less than 2 mm.
For pack size one litre to below 5 litres, the font size of the label declaration “MULTI-SOURCE EDIBLE OIL” shall not be less than 4 mm with the length of declaration statement as 45 mm minimum and for label declaration “Name and Nature of edible oil……per cent. by weight” font size shall not be less than 2.5 mm.
For pack size five litre and above, the font size of the label declaration “MULTI-SOURCE EDIBLE OIL” shall not be less than 10 mm and for label declaration “Name and Nature of edible oil……per cent. By weight” font size shall not be less than 3 mm.
There shall also be the following declaration in bold capital letters along with the name of product on front of pack –
Source: FSSAI



Sanjoy Mukhopadhyay says
Multi source oils to get rid of Agmark. FBO should allow multi source oils as per their choice subjected to better health benifits and better fatty acid profile and in different combination ratio of more than two oils ( not even third as flax seed oil ) 3 or 4 or more than 5 as per the true meaning of multi source oils.