
Through a notification in October 2013, the FSSAI had issued guidelines on Scheme of Testing and Inspection which was to be adopted by dairy processing plants for the purpose of self-monitoring and strengthening of internal controls. This Scheme of Testing and Inspection would help to ensure safe and good quality supply of milk and milk products to consumers.
FSSAI has since received several representations from various stakeholders regarding the implementation of the scheme and after consideration, scheme has been amended accordingly. The amended scheme has been given in the tables below. This revised Scheme of Testing and Inspection
- has to be followed by all dairy processing plants
- All dairy processing plants shall maintain appropriate records in this context which would be verified during surveillance visits/inspections.
SCHEME OF TESTING AND INSPECTION FOR MILK (STI)
To ensure the safety and quality of milk supplied to consumers, FSSAI has developed a scheme for sampling and testing to be implemented by dairy processing establishments to strengthen the internal controls through self-monitoring. The scheme has to be implemented at all the dairy processing establishments as per the frequency mentioned in the enclosed document. The dairy establishments shall test raw milk, in process and finished products for adulterants, hygiene indicators, microbial contaminants and safety parameters like pesticides, antibiotics, aflatoxins etc. at scheduled frequency.
The establishments shall ensure that the samplers have adequate knowledge of sampling of raw and processed milk from different locations as per the scheme. The scheme stipulates the minimum sampling points, test methods and frequency of sampling. The establishment may consider to increase the sampling points and sampling frequency as per their requirement with respect to capacity, production process and risk associated with the product and process.
All dairy processing establishments shall have a well-equipped in-house laboratory for testing microbiological and other chemical parameters. The testing shall be done by qualified and trained laboratory personnel. In case of unavailability of in-house testing facilities for test parameters that require advanced analytical equipment, the tests may be carried out at FSSAI notified lab. The details of FSSAI notified labs are available at www.fssai.gov.in
The establishments shall maintain all the test records at least for a period of one year in addition to the documents and records listed in Part 3 of Schedule 4 of Food Safety and’ Standards (Licensing and Registration) Regulations, 2011. The results of all such tests shall be made available to the officials of Food Authority or Food Safety Commissioner for verification as and when required.
- In case any non- compliance is reported and deviation can be corrected without affecting the safety and quality of milk, then it should be re-processed and again tested to ensure that re- processed milk conforms to all the requirements.
- However, if there is a non-compliance on parameters which cannot be corrected even by re-processing like presence of residues, adulterants etc., the entire batch shall be rejected. In case its already under process, then production shall be stopped immediately.
- The production shall be resumed only after carrying out root cause analysis for the non-compliance, corrective and preventive action is taken and its effectiveness verified during subsequent testing. The records of all such investigation shall be maintained.
A separate record shall be maintained giving information on quality and batch number as applicable, relating to all such rejections/defective/sub-standard material of the production not conforming to the requirements and the method of its disposal. Such material shall in no case be stored together with that conforming to the specification.
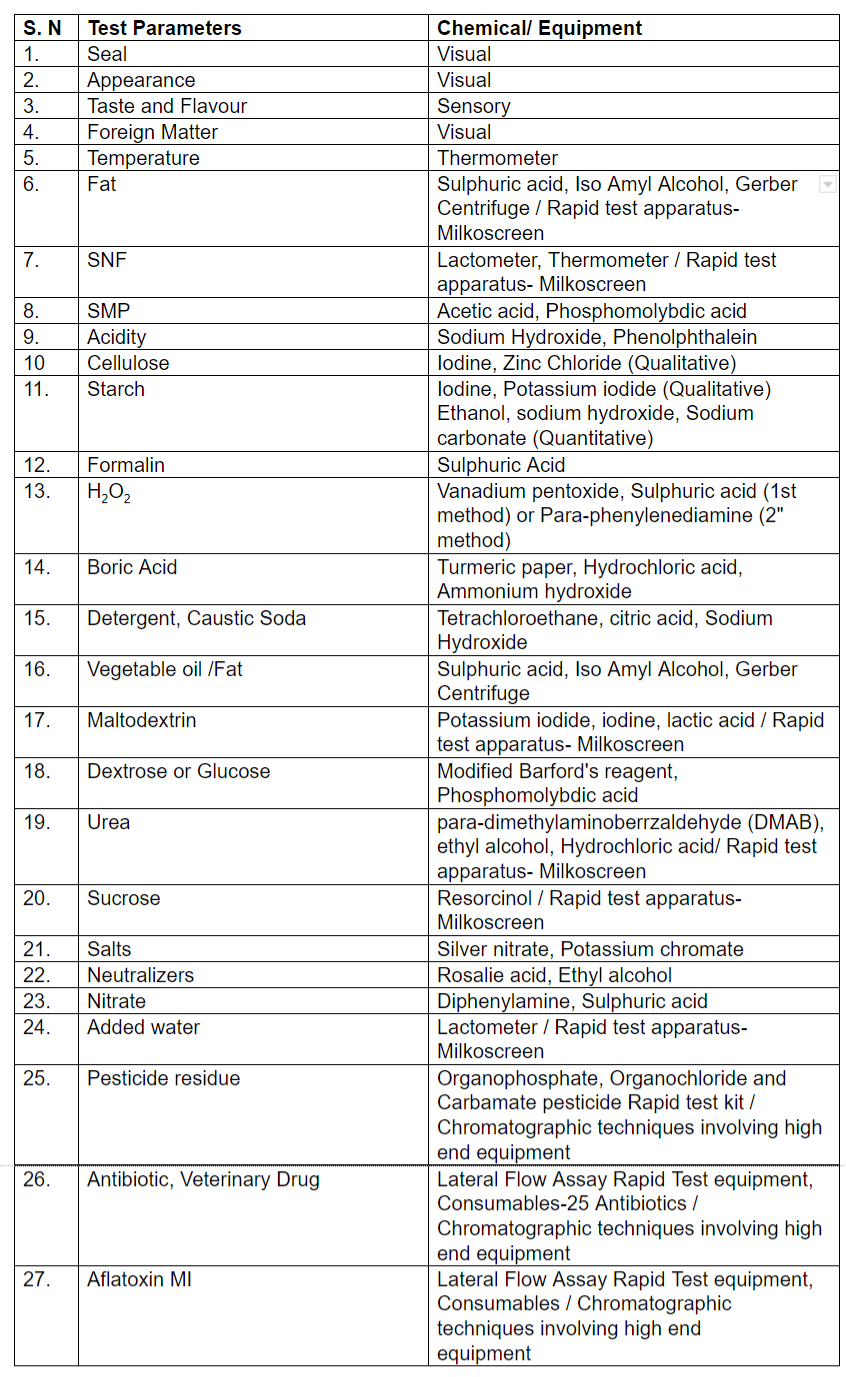
The scheme document, format of reporting and minimum lab facility/equipment required to establish in-house testing facility at raw milk reception dock is given below.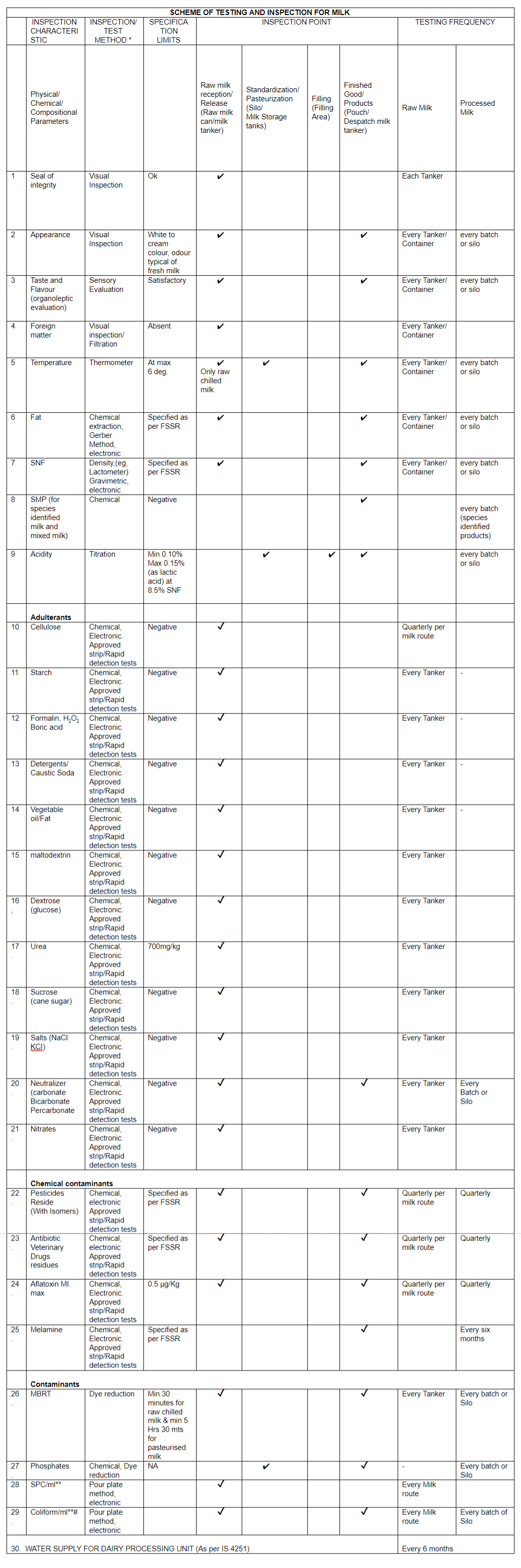 Note: “FSSAI manual of Methods of Analysis for Milk and Milk Products and any other appropriate method which includes BIS test method. FSSAI approved Rapid kit or test methods as applicable.
Note: “FSSAI manual of Methods of Analysis for Milk and Milk Products and any other appropriate method which includes BIS test method. FSSAI approved Rapid kit or test methods as applicable.
**It is only a hygiene indicator, # Desirable but the FBO could take decision on what best to be done for compliance and safety related to marked parameter.
Information required for testing of milk and milk products
Note: FSSAI Manual of Methods of Analysis for Milk and Milk Products and any other appropriate method which includes BIS test methods, AOAC test methods, FSSAI approved Rapid kit or test method as applicable
Format in which records of STI are to be maintained by dairy establishments
| S. No. | Date | Test Parameter as per STI | Test Method | Sampling Point | Batch no./Silo no./Tanker no. | Results | Action taken in case of con-comp |
Source: FSSAI
Leave a Reply